|
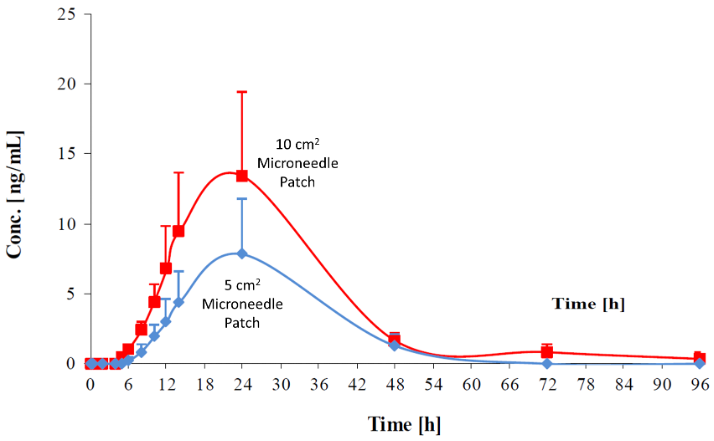
KETARX Ketamine Patch demonstrated extended delivery of ketamine over 40 hours KETARX Ketamine Patch offers flexible dosing through increased size, drug load or concentration of ketamine to better align with intramuscular and intravenous administration Initiating clinical studies by the end of Q4-2022 to support phase 2 and 3 clinical studies in depression, post-traumatic stress disorder, and pain indications Following the 505(b)(2) regulatory pathway for FDA approval of KETARX Ketamine Patch TORONTO, September 7, 2022 -- PharmaTher Holdings Ltd. (the “Company” or “PharmaTher”) (OTCQB: PHRRF) (CSE: PHRM), a specialty pharmaceutical company focused on developing and commercializing novel uses and delivery forms of ketamine to treat mental health, neurological and pain disorders, announced today that it has completed an IND-enabling pharmacokinetic and tolerability study (the “Study”) of KETARX Ketamine Patch in minipigs. The KETARX Ketamine Patch was manufactured by its CDMO partner, LTS LOHMANN Therapie-Systeme AG. The Study was completed at Aurigon Toxicological Research Center Ltd. (“ATRC”), under full Good Laboratory Practice compliance, which is a prerequisite for upcoming phase 2 clinical studies according to FDA guidelines. PharmaTher is developing KETARX Ketamine Patch to improve the administration burden in support of current ketamine use and as a potential therapeutic alternative in the treatment of various mental health, neurological and pain disorders. Fabio Chianelli, CEO of PharmaTher, commented: “This IND-enabling study supports our continued development of an alternative approach to deliver ketamine. We are in the final stage of preparing for our clinical studies with KETARX Ketamine Patch, which we believe will transform how ketamine is used to treat underserved medical conditions in mental health, neurological and pain disorders. We plan to initiate clinical studies by the end of Q4-2022 to determine pharmacokinetic parameters, dose range, dose response and tolerability that will support later phase 2 and 3 clinical studies in depression, post-traumatic stress disorder, and pain indications. We plan to follow the 505(b)(2) regulatory pathway for FDA approval of KETARX Ketamine Patch.” Actual prototype of KETARX Ketamine Patch Study Overview This Study aimed to investigate the plasma concentration and pharmacokinetics of ketamine after administration of KETARX Ketamine Patches to female Göttingen Minipigs. The degree of local tolerance was examined as reflected by erythema, edema formation and other effects (e.g. lesions) after removal of the pressure dressing. To determine the concentration of ketamine in the blood plasma, samples were collected up to 4 days after the administration. The plasma concentrations of ketamine were measured and the pharmacokinetic evaluation was performed at the Analytical Department of ATRC. Study Results The delivery of ketamine through intramuscular injection results in rapid delivery of a bolus dose of ketamine. To alter the speed and availability of ketamine, the use of intravenous infusion is the standard strategy; however, it requires complex equipment and technical expertise to administer. The Study demonstrated that KETARX Ketamine Patch applied directly to the skin was well tolerated and capable of delivering a ketamine dose over an extended period of time without modification of ketamine or additional formulation enhancement. Pharmacokinetic evaluation of intradermal administration of ketamine via KETARX Ketamine Patch demonstrated extended delivery of ketamine over 40 hours with a slow and elongated increase of plasma levels over 18 hours and a slow decline for an additional 24 hours in a linearly proportional manner relative to dose (FIGURE #1). FIGURE #1: Blood levels of ketamine delivery with KETARX ketamine patch relative to time with either a 5 cm2 or 10 cm2 patch. Overall, the small patch size used in this Study supports the opportunity to increase dose through increased size, drug load or concentration of ketamine and will allow increases in ketamine dosing to better align with intramuscular and intravenous administration. No clinical symptoms, mortality or body weight differences were observed throughout the study. Local tolerance and histopathology assessment of the patch without ketamine demonstrated that it is well tolerated and with ketamine, the patch showed only a minor difference compared to control.
About KETARX Ketamine Patch KETARX Ketamine Patch aims to deliver ketamine for intradermal administration to treat various mental health, neurological and pain disorders. It consists of hydrogel-forming microneedle arrays and an accompanying reservoir that will overcome limitations by the quantity of drug loaded into the needles or onto the needle surfaces. As such, the patch can significantly increase the amount of drug that can permeate through the microneedle array and into the skin1 and is tailored for ketamine due to the required drug volume to maximize its therapeutic utility and increase potential market opportunities. KETARX Ketamine Patch also aims to empower patients to dose their medication remotely, safely and conveniently rather than being supervised by a healthcare provider at a certified medical office, although it can also be utilized in these types of settings. It has the potential for enabling continuous delivery of ketamine (without pain) with minimal formulation manipulation into systemic circulation while maintaining constant plasma levels for more than 24 hours, which will improve efficacy and compliance for patients.1-2 Also, KETARX Ketamine Patch will incorporate anti-tampering and anti-abuse features that parallel the approach used by commercially available tamper-resistant transdermal fentanyl patches. About PharmaTher Holdings Ltd. PharmaTher Holdings Ltd. (OTCQB: PHRRF) (CSE: PHRM) is a specialty pharmaceutical company focused on developing and commercializing novel uses and delivery forms of ketamine to treat mental health, neurological and pain disorders. PharmaTher’s product portfolio consists of KETARX (ketamine) delivered by intravenous injection, intradermal microneedle patch, and subcutaneous pump administration. Learn more at PharmaTher.com. For more information about PharmaTher, please contact: Fabio Chianelli Chief Executive Officer PharmaTher Holdings Ltd. Tel: 1-888-846-3171 Email: [email protected] Website: www.pharmather.com Neither the Canadian Securities Exchange nor its Regulation Services Provider have reviewed or accept responsibility for the adequacy or accuracy of this release. Cautionary Statement This press release contains 'forward-looking information' within the meaning of applicable Canadian securities legislation. These statements relate to future events or future performance. The use of any of the words "could", "intend", "expect", "believe", "will", "projected", "estimated", "potential", "aim", “may” and similar expressions and statements relating to matters that are not historical facts are intended to identify forward-looking information and are based on PharmaTher Holdings Ltd. (the "Company") current belief or assumptions as to the outcome and timing of such future events. Forward-looking information is based on reasonable assumptions that have been made by the Company at the date of the information and is subject to known and unknown risks, uncertainties, and other factors that may cause actual results or events to differ materially from those anticipated in the forward-looking information. Given these risks, uncertainties and assumptions, you should not unduly rely on these forward-looking statements. The forward-looking information contained in this press release is made as of the date hereof, and Company is not obligated to update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as required by applicable securities laws. The foregoing statements expressly qualify any forward-looking information contained herein. Factors that could cause actual results to differ materially from those anticipated in these forward-looking statements are described under the caption "Risk Factors" in Company's management's discussion and analysis for the three and nine month periods ended February 28, 2022 and 2021 ("MD&A"), dated April 25, 2022, which is available on the Company's profile at www.sedar.com. This news release does not constitute an offer to sell or the solicitation of an offer to buy, and shall not constitute an offer, solicitation or sale in any state, province, territory or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state, province, territory or jurisdiction. Sources: 1. Donnelly R.F. Hydrogel-forming microneedles prepared from "super swelling" polymers combined with lyophilised wafers for transdermal drug delivery. PLoSONE. 2014 doi: 10.1371/journal.pone.0111547. 2. Courtenay, et al. Hydrogel-forming microneedle arrays as a therapeutic option for transdermal esketamine delivery, Journal of Controlled Release, Volume 322, 2020, Pages 177-186. Comments are closed.
|
Archives
May 2024
Categories |


 RSS Feed
RSS Feed

9/7/2022